Circadian Rhythms, Chronodisruption & the Microbiota: Timing is Everything
Are the trillions of microorganisms in the human gut sensitive to time? It is safe to say that many human biological processes have cycles of time, or “clocks” associated with them. Biological rhythms may follow a day/night cycle, a 24-hour cycle, a monthly cycle, or even an annual cycle. Researchers studying digestion and the microbiota have run across ample evidence of 24-hour cycle (circadian rhythm) disruptions causing physical changes, as well as changes in behavior and cognition. Indeed, circadian rhythms (CRs) affect multiple aspects of human digestion: absorption, motility in the gut, effects on hormones and gut barrier function, and the disruption of gut microbiota (1). Since we all eat and sleep, the odds for disruption of some sort are high.
The ability of a disturbed biological clock, such as the 24-hour circadian rhythm, to negatively affect a biological process to the point of causing a chronic issue has been given a name: chronodisruption. The term “chronodisruption” was forwarded by researchers Erren and Reither in 2003 to distinguish it from “circadian disruption” or “disruption of circadian rhythms,” which they deemed as less severe, temporary, and not inevitably ending in chronic processes (2).
Interest in chronodisruption is starting to spread as increasing numbers of studies show feedback loops encompassing metabolic factors, circadian rhythms, and the microbiota. Circadian rhythm disruption has been associated with chronic illnesses such as cancer, cardiovascular disease, diabetes, and obesity. Furthermore, disrupted timing cycles can affect the immune system and pique inflammation. Chronodisruption is merely the extreme end of consistently disrupted circadian rhythms, resulting in chronic processes.
There is no ready figure for the number of people experiencing chronodisruption. However, if one looks at the evidence for circadian rhythm disruption, estimates of apparently sleep-deprived individuals are quite high. Roughly a third of the adult U.S. population reports getting less sleep than is recommended for their age group (3). It is unknown how many individuals experience jet lag from traveling and “social jet lag,” where there is a mismatch between the body’s biologically determined preferred sleep time and social obligations (4), but these situations represent circadian disruption as well. Many things that regularly disturb the circadian rhythms eventually result in chronodisruption, including the well-known disturbance from light, as well as the lesser-known eating out of regular feeding times, called “mistimed eating” (5). Analyzing the various factors in this dynamic is worth examination, not only to be a tale of caution but to highlight the need for lifestyle interventions.
A Brief Review: Processes Affected by Circadian Rhythms
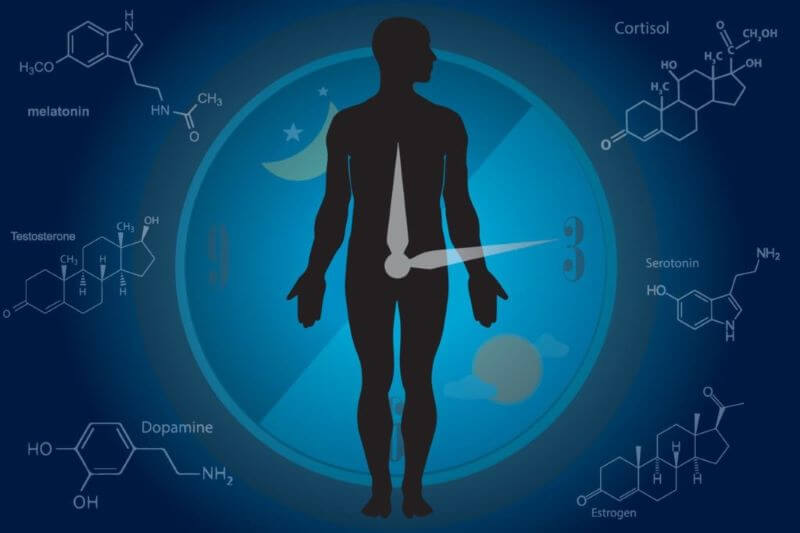
Many human biological processes are affected by or appear regulated by circadian rhythms. The sleep-wake cycle is probably the most obvious, which encourages sleep when it is dark and wakefulness when it is light outside. A variety of hormones appear quite attuned to the circadian rhythm, including melatonin, growth hormone, and cortisol. There is also evidence of circadian rhythms affecting blood pressure and body temperature. The immune system seems affected as well. Even complex metabolic processes such as digestion show signs of influence from daily cycles.
How We Keep Time – What Is the Physiological Basis for Clocks?
The master clock for the human has been located in a certain group of cells called the Suprachiasmatic Nucleus (SCN), which are part of the hypothalamus. This special group of cells is regulated by environmental cues, such as light and dark cycles, and temperature variations, and by internal cues such as hormones. The main function of this master clock is to synchronize internal clocks with these cues from the environment, which ultimately affect a variety of processes. Light entrains the master clock by stimulating photo-sensitive cells, which convert those signals to neurotransmitter substances that affect other cells. Eventually, the neurotransmitter substances emitted entrain the hypothalamic-pituitary-adrenal (HPA) axis, leading to the secretion of glucocorticoids that follow circadian oscillations (6). Glucocorticoids go on to influence cognition, the body’s responses to stress, and various aspects of metabolism and the immune system.
Peripheral clocks are any clock outside of the SCN, located throughout the body but also in the brain (some are found in the hippocampus and amygdala). They help control circadian, seasonal, and annual cycles of rest and activity. Peripheral clocks are also responsive to light, temperature, and feeding times (7).
There are many different types of clocks, and more are being discovered by the day. Already identified are circadian, diurnal, ultradian, infradian, tidal, lunar, seasonal, and annual clocks. Cellular clocks are an exciting, evolving field and a subject for another time.
Metabolic Factors that Contribute to Circadian Rhythm (CR) Disruption
There are a variety of metabolic factors that can affect the microbiota through disrupted circadian rhythms. Due to their prevalence in the US population, some of the more interesting metabolic factors are impaired circadian rhythms, changes in nutrient availability, and changes in hormones from late-night eating.
Disrupted Circadian Rhythms:
It seems circular or perhaps a case of “stating the obvious,” but circadian rhythms are metabolic in nature, as the body clock is influenced by the body’s metabolism, among other factors. The body’s metabolism is regulated by hormones and other biochemical processes that are influenced by the body’s internal clock. This internal clock is responsible for controlling various metabolic processes such as hormone secretion, energy production, and the timing of food breakdown. As a result, circadian rhythms are closely related to the body’s metabolic processes and readily affect body function.
Circadian rhythms can be disrupted by stress, jet lag, shift work, changes in cycles of light and dark, sleep disturbances, and eating habits. Medications, caffeine, and timing or amount of exercise may also disrupt circadian rhythms. Regulated by the hypothalamic-pituitary-adrenal (HPA) axis, circadian rhythms in turn affect the production of hormones such as growth hormones, melatonin, and cortisol.
There is a fair amount of influence over the gastrointestinal (GI) system through circadian rhythms. Specifically, diurnal rhythms, which are the cycles of activity during the day and rest at night, and they greatly affect digestion, absorption, gut barrier function, gut microbiota, and gut motility (1).
Changes in Nutrient Availability:
Changes in nutrient availability can contribute to the disruption of CRs by altering body metabolism.
- High Fat Diet – A high-fat diet can increase the production of free radicals, causing oxidative stress, cellular damage, and inflammation. This can disrupt the body’s natural circadian rhythm, leading to chronic constipation as one possible result. In addition, a high-fat diet can increase the production of hormones such as cortisol, which can also disrupt the body’s natural circadian rhythm. Finally, a high-fat diet increases the production of inflammatory molecules that may also contribute to impaired CR (8). All of these metabolic changes can cause a general disruption of the body’s circadian rhythms.
Temporary imbalances in the microbiota occur when a high-fat diet is consumed. This can lead to an imbalance in the overall composition of the microbiota, resulting in reduced beneficial bacteria and increased pathogenic bacteria. For example, a high-fat diet can lead to an increase in the bacteria phylum Firmicutes and a decrease in Bacteroides, which can lead to an increased risk of chronic diseases, inflammation, and obesity (9).
- Reduction of Fiber – Fiber is a primary food source for beneficial bacteria in the gut. A restriction of fiber in the diet is associated with lower numbers of beneficial bacteria in the phylum Bifidobacteria (10), leading to increased inflammation and metabolic disorders (11).
Short-chain fatty acids (SCFAs) are produced by the fermentation of fiber in the large intestine. SCFAs are important for maintaining a healthy microbiota, acting as an energy source for intestinal epithelial cells to control the growth of healthy bacteria and help regulate the immune system. Low-fiber diets restrict SCFA activities, including reducing inflammatory cytokines and playing a part in gut-brain communication (endocrine and immune signaling pathways) (12).
To make matters worse, a low-fiber diet can lead to the growth of opportunistic pathogens such as Clostridium difficile, which can cause severe diarrhea and other painful gastrointestinal symptoms.
Changes in Hormone Production from Late-Night Eating:
Hormones, especially melatonin and cortisol, play an important role in regulating the body’s internal clock. Melatonin levels rise and fall in response to light and darkness, while cortisol levels rise and fall in response to stress and other external influences. Changes in hormone production disrupt the body’s hormones, causing menstrual disorders. This disruption can lead to a variety of health problems, including sleep disturbances, changes in appetite, mood swings, and disruption of the body’s natural circadian rhythms. Hormones can be disrupted by a wide array of things. However, late-night eating gets a notable mention because of its prevalence in American culture.
All of the following changes in hormone production due to late-night eating can lead to circadian rhythm disruption, resulting in imbalanced or damaged microbiota and an increased risk of disorders of digestion.
- Eating at night inhibits the production of melatonin, a hormone that helps regulate the body’s sleep-wake cycle. Melatonin levels rise and fall in response to light and darkness, providing a significant cue to circadian rhythms. Most people don’t eat in the dark. Eating late at night exposes the eyes to light, inhibiting the production of melatonin, which should peak during the hours of sleep. This inhibition can cause changes in the production of other hormones, such as ghrelin and cortisol.
Cortisol levels rise and fall in response to the influence of melatonin circadian rhythms, stress, and other external influences. High cortisol levels can interfere with the growth of bacteria in the microbiota, harming gut mucosa and increasing inflammation (13).
- Eating at night inhibits the production of leptin, a hormone that helps regulate hunger and satiety. Low levels of leptin can be seen for 24 hours after late-night eating, disrupting its natural cycle (14). The body’s natural hunger signals get confused, causing overeating and an imbalance in the production of other hormones such as ghrelin and insulin. Overeating, of course, can damage the delicate balance of microbes in the microbiota. Additionally, peripheral clocks are negatively affected by “mistimed eating” (5), which can happen easily if hunger signals get confused, so inhibited leptin production can reinforce disrupted circadian rhythms.
- Eating late at night can also interfere with the production of the appetite-stimulating hormone ghrelin. When food is eaten, ghrelin, which is produced in the stomach, is suppressed. This can delay the natural cycle of ghrelin. When ghrelin production is disrupted, the body’s natural hunger signals become confused, which can cause overeating and an imbalance in the production of other hormones, such as insulin and leptin.
Ghrelin levels also affect gastrointestinal (GI) motility, which is one way it can ultimately affect the microbiota. GI motility helps stir and push the microbiota in the gut, helping to maintain a good balance of bacteria. Gut motility, in turn, can affect circadian rhythms (15).
- Eating late at night can interfere with the production of insulin, a hormone that helps regulate blood sugar levels. Blood sugar levels can slowly rise after eating late at night, peaking then dipping strongly during a sleep cycle. The more sugary the meal, the more dramatic the peaks and dips will be. Many people awaken upon the blood sugar dip. The disruption of the sleep cycle can affect CRs. Also, insulin can affect the production of other hormones, such as leptin and ghrelin. Leptin helps regulate energy in the body, and an increase in insulin can cause a decrease in leptin, leading to hunger and cravings.
Sugars (which cause insulin to rise) can also affect the microbiota directly. Yeasts particularly enjoy the remnants of a high-sugar diet, and quickly overgrow in a consistently sugary medium. This situation can lead to dysbiosis, which in turn can disrupt CRs (16).
Chronodisruption: The Extreme End of the Spectrum
It is easy to notice that one hormone affects another, disrupting the circadian rhythms and the microbiota, which then negatively affects the production of other hormones. When this feedback loop continues for long periods of time, chronic processes are formed and the definition of chronodisruption is met. Inflammation figures prominently and is a likely factor in the transition from merely temporarily disrupted circadian rhythms to chronodisruption. Indeed, many of the outcomes of chronodisruption are highly associated with inflammation: cancer (17), cardiovascular disease (18), diabetes (19), and obesity (20).
Considering the microbiota’s role in the gut-brain axis, it is not wholly surprising that it, too, might be impaired by disruptions of circadian rhythms. Matenchuk, et. al. found the gut microbiota and the metabolites it produces to have diurnal rhythmicity that responds primarily to the feeding and fasting cycles (21). No doubt the composition, that is, the ratios of phyla of bacteria to each other in the microbiota, can influence this, and researchers will soon have more details on this information.
The Need for Lifestyle Interventions
What is apparent now, though, is the ability of lifestyle changes to address the chronodisruption, or better yet, prevent it from happening in the first place. The issues themselves point to the remedies. It’s the common sense advice that has been circulating for decades: “Get good sleep. Eat right. Exercise.”
To refine that advice for the current era, allowing sunlight into the eyes around the same time of day, every day, could help sync internal clocks. Taking a walk while taking sunshine into the eyes would help burn and utilize some blood sugar. Refraining from eating in the few hours before bedtime could prevent disruptions to many important hormones, and regular eating times during the day could help entrain the peripheral clocks. Eating just enough healthy fats and reducing carbohydrates (especially sugars) could go a long way towards helping the body’s metabolism stay on this side of diabetes and foster a healthy microbiota. It appears that human bodies prefer a schedule, all the way down to the trillions of microorganisms in our guts.
Sources
- Segers A, Depoortere I. Circadian clocks in the digestive system. Nat Rev Gastroenterol Hepatol. 2021 Apr;18(4):239-251. doi: 10.1038/s41575-020-00401-5. Epub 2021 Feb 2. PMID: 33531700.
- Defining chronodisruption – erren – 2009 – wiley online library [Internet]. [cited 2022Dec11]. Available from: https://onlinelibrary.wiley.com/doi/10.1111/j.1600-079X.2009.00665.x
- Adults – sleep and sleep disorders [Internet]. Centers for Disease Control and Prevention. Centers for Disease Control and Prevention; 2022 [cited 2022Dec11]. Available from: https://www.cdc.gov/sleep/data-and-statistics/adults.html
- Jankowski KS. Social jet lag: Sleep-corrected formula. Chronobiol Int. 2017;34(4):531-535. doi: 10.1080/07420528.2017.1299162. Epub 2017 Mar 20. PMID: 28318321.
- Challet E. The circadian regulation of food intake. Nat Rev Endocrinol. 2019 Jul;15(7):393-405. doi: 10.1038/s41574-019-0210-x. PMID: 31073218.
- Li Y, Androulakis IP. Light entrainment of the SCN circadian clock and implications for personalized alterations of corticosterone rhythms in shift work and jet lag [Internet]. Nature News. Nature Publishing Group; 2021 [cited 2022Dec11]. Available from: https://www.nature.com/articles/s41598-021-97019-7
- Schibler U, Ripperger J, Brown SA. Peripheral circadian oscillators in mammals: time and food. J Biol Rhythms. 2003 Jun;18(3):250-60. doi: 10.1177/0748730403018003007. PMID: 12828282.
- Kim SM, Neuendorff N, Alaniz RC, Sun Y, Chapkin RS, Earnest DJ. Shift work cycle-induced alterations of circadian rhythms potentiate the effects of high-fat diet on inflammation and metabolism. FASEB J. 2018 Jun;32(6):3085-3095. doi: 10.1096/fj.201700784R. Epub 2018 Feb 5. PMID: 29405095; PMCID: PMC5956251.
- Murphy EA, Velazquez KT, Herbert KM. Influence of high-fat diet on gut microbiota: a driving force for chronic disease risk. Curr Opin Clin Nutr Metab Care. 2015 Sep;18(5):515-20. doi: 10.1097/MCO.0000000000000209. PMID: 26154278; PMCID: PMC4578152.
- Simpson HL, Campbell BJ. Review article: dietary fibre-microbiota interactions. Aliment Pharmacol Ther. 2015 Jul;42(2):158-79. doi: 10.1111/apt.13248. Epub 2015 May 24. PMID: 26011307; PMCID: PMC4949558.
- Schroeder BO, Birchenough GMH, Ståhlman M, Arike L, Johansson MEV, Hansson GC, Bäckhed F. Bifidobacteria or Fiber Protects against Diet-Induced Microbiota-Mediated Colonic Mucus Deterioration. Cell Host Microbe. 2018 Jan 10;23(1):27-40.e7. doi: 10.1016/j.chom.2017.11.004. Epub 2017 Dec 21. PMID: 29276171; PMCID: PMC5764785.
- Dalile B, Van Oudenhove L, Vervliet B, Verbeke K. The role of short-chain fatty acids in microbiota–gut–brain communication [Internet]. Nature News. Nature Publishing Group; 2019 [cited 2022Dec11]. Available from: https://www.nature.com/articles/s41575-019-0157-3
- Oroian BA, Ciobica A, Timofte D, Stefanescu C, Serban IL. New Metabolic, Digestive, and Oxidative Stress-Related Manifestations Associated with Posttraumatic Stress Disorder. Oxid Med Cell Longev. 2021 Dec 20;2021:5599265. doi: 10.1155/2021/5599265. PMID: 34966477; PMCID: PMC8712172.
- Kira Sampson | Brigham and Women’s Communications. Late-Night Eating Impact [Internet]. Late-Night Eating Impact | Harvard Medical School. 2022 [cited 2022Dec11]. Available from: https://hms.harvard.edu/news/late-night-eating-impact
- Duboc H, Coffin B, Siproudhis L. Disruption of Circadian Rhythms and Gut Motility: An Overview of Underlying Mechanisms and Associated Pathologies. J Clin Gastroenterol. 2020 May/Jun;54(5):405-414. doi: 10.1097/MCG.0000000000001333. PMID: 32134798; PMCID: PMC7147411.
- Leone V, Gibbons SM, Martinez K, Hutchison AL, Huang EY, Cham CM, et al. Effects of diurnal variation of gut microbes and high-fat feeding on host circadian clock function and metabolism [Internet]. Cell Host & Microbe. Elsevier; 2015 [cited 2022Dec11]. Available from: https://www.cell.com/cell-host-microbe/fulltext/S1931-3128(15)00123-7
- Almaida-Pagan PF, Torrente M, Campos M, Provencio M, Madrid JA, Franco F, Morilla BR, Cantos B, Sousa PA, Madrid MJM, Pimentao J, Rol MÁ. Chronodisruption and Ambulatory Circadian Monitoring in Cancer Patients: Beyond the Body Clock. Curr Oncol Rep. 2022 Feb;24(2):135-149. doi: 10.1007/s11912-021-01158-z. Epub 2022 Jan 21. PMID: 35061192; PMCID: PMC8857092.
- Garcia-Rios A, Ordovas JM. Chronodisruption and cardiovascular disease. Clin Investig Arterioscler. 2022 Jun;34 Suppl 1:S32-S37. English, Spanish. doi: 10.1016/j.arteri.2021.12.004. Epub 2022 Feb 4. PMID: 35125247.
- la Fleur SE, Kalsbeek A. Increased risk of diabetes due to obesity: Does Chronodisruption play a role? [Internet]. SpringerLink. Springer New York; 1970 [cited 2022Dec11]. Available from: https://link.springer.com/chapter/10.1007/978-1-4614-5082-5_7
- Vitale JA, Lombardi G, Weydahl A, Banfi G. Biological rhythms, chronodisruption and chrono-enhancement: The role of physical activity as synchronizer in correcting steroids circadian rhythm in metabolic dysfunctions and cancer. Chronobiol Int. 2018 Sep;35(9):1185-1197. doi: 10.1080/07420528.2018.1475395. Epub 2018 Jun 28. PMID: 29953265.
- Matenchuk BA, Mandhane PJ, Kozyrskyj AL. Sleep, circadian rhythm, and gut microbiota. Sleep Med Rev. 2020 Oct;53:101340. doi: 10.1016/j.smrv.2020.101340. Epub 2020 May 13. PMID: 32668369.